
MERIT’S CONTINUED COMMITMENT TO OUR OEM PARTNERS
As the public health response to the novel coronavirus, known as COVID-19 continues, our thoughts remain with the communities and individuals severely affected by this disease. Merit Medical is fully committed to doing our part to contain the spread of COVID-19, while providing the much-needed support to healthcare professionals, hospitals, and medical device manufacturers on the front-lines of this pandemic. As always, Merit Medical OEM is devoted to maintaining the uninterrupted supply of Merit’s products and components for our OEM partners across the globe.
We are confident in the sound foundation that Merit has strategically built over the last three decades. This sound foundation gives Merit the ability to adapt and overcome the challenges we face. Here’s an overview of the precautions Merit is taking to ensure our OEM partners that manufacturing operations and delivery remain uninterrupted and that ample product supply is available.
Merit’s Current Status and Facts:
- Merit remains fully operational and firing on all cylinders.
- Merit’s critical business functions are protected by Merit’s pre-established business continuity plan.
- Merit has implemented a comprehensive COVID-19 response plan covering all of Merit’s global facilities.
- Merit’s Global COVID-19 Task Force meets daily to stay on top of this dynamic situation, ensuring Merit’s response plan includes the most current information available.
- Our primary mission is to provide the uninterrupted supply of Merit’s quality products.
- Merit OEM is committed to providing attentive customer service and helping our OEM partners establish joint COVID-19 response plans.
BUSINESS CONTINUITY PLAN
Supply Chain Management
Merit has worked hard to establish a secure supply chain that has not been impacted by the COVID-19 outbreak. Should any disruptive circumstances develop beyond our current knowledge, Merit’s OEM Partners will be promptly informed.
Safety Stock and Dual Sourcing of Materials
Merit mitigates supply chain risk with appropriate safety stocks and dual sourcing of materials. Merit’s procurement and planning teams are vigilantly monitoring the situation for changes. We are confident in our supply chain and do not anticipate any shortages.
Vertical Integration
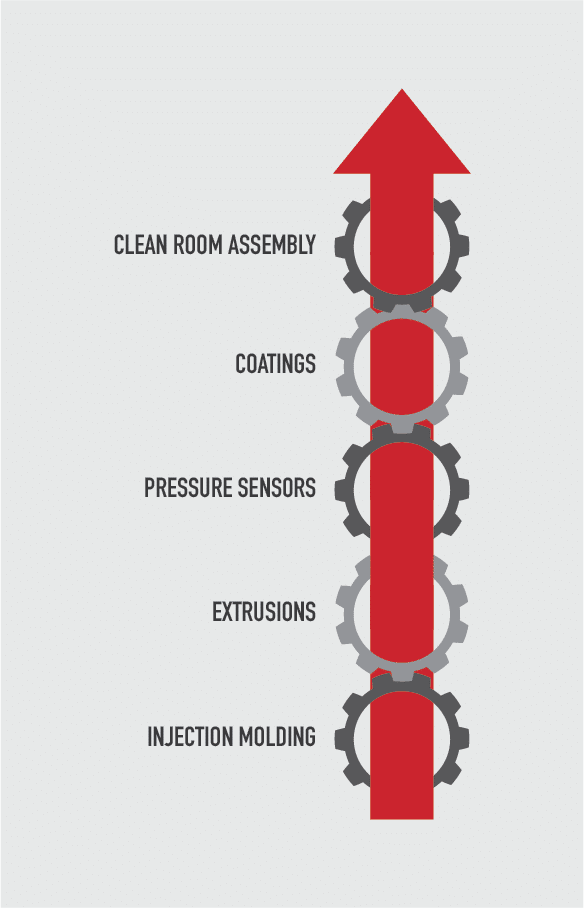
Merit’s strategy from inception was to be as vertically integrated as reasonable to avoid disruptions. To that end, Merit has developed manufacturing capabilities such as injection molding, extrusions, coatings, sensors, and clean room assembly that are now the core of our business.
COVID-19 PRECAUTIONS

Personal Protective Equipment
- All personnel entering any of Merit’s global facilities must wear personal protective masks.
- Merit is providing PPE for all essential employees to help protect our workforce and products.

Separation Barriers
- Separation barriers have been installed in all of Merit’s global facilities in areas where social distancing protocols cannot be maintained.
- Some examples of these locations include cafeterias, hand washing stations, cleanroom assembly lines and cleanroom workstations.

Increased Hygiene and Cleaning
- Implemented increased hygiene measures under recognized standards.
- Posting notices to employees regarding proper hygiene.
- Increased the rate at which cleaning crews disinfect surfaces and clean facilities.

Remote Workforce
- Limit potential exposure and the spread of the virus to essential production workers and operational staff.
- Keeping Merit OEM engineers, customer service, and sales representatives active in their roles and safe in their remote offices.

Social Distancing Protocals
- Adjustments to common areas and workstations to follow social distancing guidelines.
- Eliminating group gatherings
- Requiring staff to eat in the same groups they work with.

Mandatory Medical Screening
- Merit will conduct medical screening with all staff before entering all Merit’s facilities each day.
- Merit has also providing additional leave in some circumstances to encourage employees to report when they are ill.

Travel Restrictions
- Restricting all employee travel
- Requiring self-quarantine after certain personal travel
- Screening employees individually when they have potentially been exposed to COVID-19.
- In place of face-to-face meetings, we will leverage cloud-based communication platforms to conduct business, collaborate, and further our business relationships with our partners.
Essential Life-Sustaining
- All Merit Medical facilities are engaged as a critical infrastructure business and are exempt from “forced shutdown” or “shelter in place” orders.
- Suppliers critical for Merit to fulfill its mission as an essential life-sustaining business, fall within the exemption from emergency orders under federal and state law.
- Gov. Tom Wolf of Pennsylvania ordered all non-life sustaining businesses in the state to close their physical locations.
- Merit’s manufacturing facility in Malvern, PA is classified as an essential life-sustaining business and will continue to manufacture products as scheduled.

Closed Campus Policy
- Merit Medical has closed all global facilities to all visitors, vendors, and customers.
- All on-site audits scheduled through the end of Q2 have been rescheduled during Q3 or Q4.

Continued-Improvement
- Dr. Nicole Priest, Chief Wellness Officer for Merit Medical continues to improve safety measures to ensure Merit’s workforce stays healthy.
- Merit is continuing to implement the latest guidelines from the following organizations:
MERIT’S COVID-19 SAMPLE COLLECTION AND TRANSPORT KIT
Merit Medical announced on April 21, that it has commenced production of a sample collection and transport kit, comprised of a nasopharyngeal swab and transport vial, used to collect specimens with suspected presence of Coronavirus Disease 2019 (COVID-19). The kit will be produced at Merit’s facility in South Jordan, Utah, with other Utah companies providing various services.
For the last several weeks, officials from Merit and the State of Utah have been working together in anticipation of an impending swab shortage.
“We have directed resources to the development of this kit, and our engineers, technicians, marketers and production staff have responded by working tirelessly to bring this important product to market in 30 days,” said Fred P. Lampropoulos, Merit’s Chairman and CEO. “We are especially pleased to provide this kit at a time when testing is an essential element of combatting the COVID-19 pandemic. We are increasing our tooling and production capacity to meet future anticipated demand.”




