NOT ALL SYRINGES ARE CREATED EQUAL
FDA’s Investigation into Syringes Manufactured in China
In light of recent developments regarding the FDA’s investigation of syringes manufactured in China, we want to take this opportunity to reaffirm our unwavering commitment to manufacturing syringes in the United States with unrivaled quality and reliability.
On March 18, 2024, the FDA issued warning letters to multiple manufacturers in China, citing various violations. Furthermore, the FDA’s safety communication highlighted that issues with syringes made in China are more widespread than previously known, prompting a recommendation for immediate transition away from such products.
Recent national news coverage has shed light on a broader quality concern, encompassing more syringe manufacturers in China, citing multiple class one recalls due to syringe quality issues coming from China. This heightened scrutiny underscores the critical importance of ensuring product safety, reliability, and selecting the right syringe supplier.
Merit Medical OEM is globally recognized as a trusted supplier and strategic partner, providing comprehensive product development support and the essential source of the highest quality polycarbonate syringes. We take pride in our meticulous manufacturing processes and rigorous quality control measures. For over three decades, we have been dedicated to delivering the highest quality accessory products and components to our OEM partners, who provide clinicians worldwide with life-saving tools.
Our polycarbonate syringes are molded and assembled in the United States, adhering to the strict standards of our ISO 13485 certified quality management system. All our syringes hold 510(k) clearance, and we have updated our regulatory filings in the EU to obtain a CE mark under MDR for both sterile and bulk non-sterile syringes.
Merit understands that not all syringes are equal, so we have made it our mission to manufacture syringes with unrivaled quality, performance, and reliability.
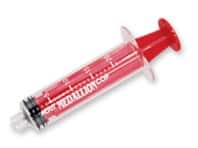
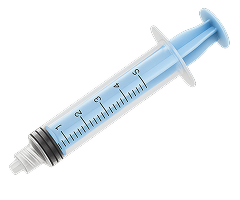
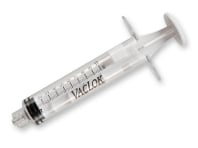
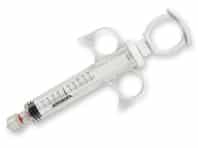
Discover Merit’s Complete Line of Polycarbonate Syringes
Customizing Merit’s Proven Products
Partnering with Merit OEM means complete access to Merit’s Core Products. Customize one of Merit’s proven products to meet your specific requirements. Syringe customization options include:
- Custom Plunger Colors
- Custom Pad Printing
- Mold Modification

Custom CE Marked Sterile Syringes
Working with Merit OEM provides you with Merit’s regulatory, legal, engineering, and manufacturing capabilities to bring a MDR certified sterile syringe to the EU market. Setting up your private labeled sterile syringe with Merit OEM includes:
- Updated labeling according to MDR requirements
- Private label formats for unit, carton, and shipper
- Custom part number specific for your private labeled syringe

REQUEST A VIRTUAL MEETING WITH MERIT’S PRODUCT DEVELOPMENT TEAM
Toll-Free: +1 800 637 4839
Phone: +1 801 208 4313
Hours: 8 am to 5 pm MST | Mon-Fri