
ISO 80369-7:2021 &
MERIT’S PLANS FOR COMPLIANCE
Increasing Patient Safety with Non-Compatible Luers
At Merit’s foundation is the desire to make innovative products, that are safe for the patient by design. The ISO 80369 series of standards developed by The International Organization for Standardization (ISO) was created with that same desire. ISO aimed to develop an innovative series of standards that created inherent patient safety through incompatible connectors used in intravascular, breathing systems, enteral, urethral/urinary, cuff inflation, and neuraxial applications. Once implemented, ISO 80369 removes the ability to make inappropriate connections or misconnections in a clinical setting through non-compatible connection groups called out in ISO 80369.

The New Luer Standard for Intravascular and Hypodermic Applications
The new luer standard ISO 80369-7 for intravascular applications has the largest impact on Merit’s product catalog. The first version of this standard was published in 2016 with the designation ISO 80369-7:2016. After the standard was published, Merit committed substantial resources towards planning and executing a proactive plan for compliance.
Merit’s initial timeline for compliance was to transition all affected products to ISO 80369-7:2016 by the end of 2020, ahead of the original October 2021 date recommended by ISO. Merit also identified some products as “priority” with a quicker compliance timeline.
During Q4 of 2019, ISO withdrew ISO 80369-7:2016 and began developing the second version of the standard. This action resulted in the FDA removing the withdrawal date of ISO 594-1/2 and announced the continued acceptance of ISO 594-1/-2 and ISO 80369-7:2016 as consensus standards.
At this point, Merit was aggressively pursuing compliance, having already transitioned some of our “priority” products from ISO 594-1/2 to ISO 80369-7:2016. However, the actions taken by ISO and FDA resulted in Merit reevaluating the speed at which this compliance project would be pursued.
After months of anticipation, ISO 80369-7:2021 is now published. Merit’s cross-functional compliance team is now reengaged with this project. Recognizing the importance of this standard, Merit is committed to transition all relevant products from ISO 594-1/2 to ISO 80369-7:2021.
Following the publication of ISO 80369-7:2021, Merit received information about the EU’s plan to harmonize ISO 80369-7:2021 to MDR. The tentative timeline for harmonizing 80369-7 to MDR is scheduled for the middle of Q2 2024. After the standard is officially harmonized by the EU, products seeking a CE mark will be required to comply with ISO 80369-7:2021 under MDR.
Merit’s ISO 80369-7:2021 Compliance Project
ISO 80369-7:2021 cancels and replaces the first version of the standard published in 2016. The revisions included in ISO 80369-7:2021 clarify some requirements to facilitate the implementation and certification of this standard.
Most of the standard has stayed the same, allowing Merit to preserve our original compliance strategy. Merit will continue to use our state-of-the-art CT scanning “Vision System” for dimensional testing. This precision equipment allows Merit to confirm the dimensional requirements have been met. Other testing methods required by the ISO 80369-7:2021 include stress cracking, fluid leakage, sub-atmospheric air leakage, axial load, unscrewing (lock luers only), overriding (lock luers only).


Considering ISO 80369-7:2021 didn’t expand the requirements of the previous version, Merit’s ISO 80369-7:2016 compliant products will now be compliant to be ISO 80369-7:2021 without the need to retest for functional or dimensional compliance.
Availability of Compliant Products
The design modifications and new testing protocols needed for each specific part number to comply with ISO 80369-7:2021 are extensive. Merit is actively working towards compliance on our list of “priority” products. Transitioning products to ISO 80369-7:2021 will follow Merit’s quality management system protocols and approvals. The changes needed for compliance for a specific part number will be completed after the engineering change notification has been approved by all responsible parties.
Merit keeps on-hand inventory of our sub-assembly molded components and products. This allows Merit to reduce lead times and helps us control our manufacturing cost. Some products and components, included in this compliance project, have on-hand inventory manufactured prior to completing the updates for ISO 80369-7. This on-hand inventory needs to be exhausted before manufacturing new products and components that include the updates for ISO 80369-7 compliance.
For more information about timelines on ISO 80369-7 updates and when compliant products and components will be available, please contact your Merit OEM Business Development Manager or your Merit OEM Customer Service Representative at oemorder@merit.com.
Compatibility with 594-1/2
In general terms, 594-1/2 was used as a basic framework for many of the dimensions and requirements called out in 80369-7. Being part of the ISO 80369 series, 80369-7 also includes non-interconnectable characteristics and tighter tolerances that ensure non-compatibility with other types of small bore connectors. The close relation between these two standards makes 80369-7 compliant luers backwards compliant with 594-1/2. Conversely, 594-1/2 luers that pass functional testing will be compatible with 80369-7 compliant luers. These two standards are 100% compatible with each other.
Requirements for Compliance
Some of the new requirements included in ISO 80369-7 that were not required in the previous luer standard, ISO
594-1/2, are listed in the table below. After the brief summary of the new requirement, we’ve listed Merit’s approach to compliance for that specific requirement.
Merit recommends reviewing the standard for a comprehensive summary of new requirements. You can access the published standard on ISO’s website at www.iso.org.
New with ISO 80369-7
Merit’s Plan to Meet the New Requirements
ISO 80369-7 includes additional functional testing requirements.
Merit will perform single sample functional testing on each luer type (slip male, slip female, threaded male, threaded female, rotating male) for each resin type as described in the master protocol.
New dimensional requirements Include:
- ISO 594-1/2 luers didn’t need to meet dimensional requirements of the standard, if the luer connection passed functional testing. ISO 80369-7 requires compliance with both the dimensional and functional requirements.
- Dimensional requirements must be met for all REQUIRED dimensions included in the standard, regardless of luer connection passing functional testing.
- Separate test methods for functional and dimensional compliance
- Testing with gauges IS NOT included in testing protocol
- Some dimensions have tolerances that previously where not included
- Dimensional testing will be performed by a new state-of-the-art VISION SYSTEM, purchased specifically to meet the stringent and rigorous testing protocol requirements of this standard.
- Merit will be verifying measurements and tightening tolerances to meet the standard. Mold modifications might be needed for some part numbers to meet dimensional requirements and increased tolerances.
- Merit will perform separate dimensional and functional testing required by the standard to ensure compliance.
Conditioning prior to functional testing is required
Merit will conduct dimensional and functional testing at T=0 and T=life of the product
Inner male lumens CANNOT EXCEED 8.3F or 0.114” and be compliant to 80369-7
- Merit OEM offers a comprehensive line of valves, including valves with inner lumen smaller than 8.3F
- Merit will not seek compliance on devices designed specifically with a LARGE BORE inner lumen.
Dimensional Requirements
Merit will review and update our luer connection to include tightened tolerances and dimensional requirements specified in the standard. The following information describes some of the design changes Merit will make for ISO 80369-7:2021.
Merit recommends reviewing the standard for a comprehensive summary of dimensional requirements. You can access the published standard on ISO’s website at www.iso.org.
Male Luer
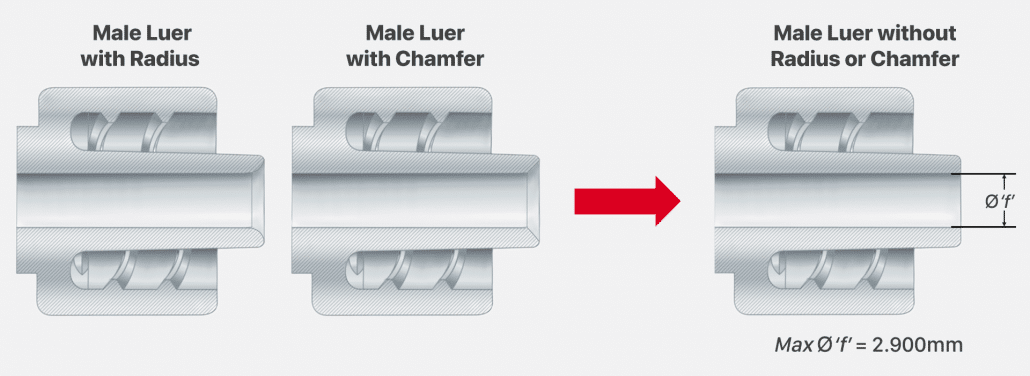
Some of Merit’s products feature a male slip luer or a male lock luer designed with a chamfer or a radius to the inner dimension of the luer. Male luers with this chamfer or radius have an inner dimension that exceeds the max ‘f’ dimension of 2.900mm (0.114”). Removing the chamfer or radius will allow Merit’s male luer to meet the required ‘f’ dimension and comply with ISO 80369-7:2021.
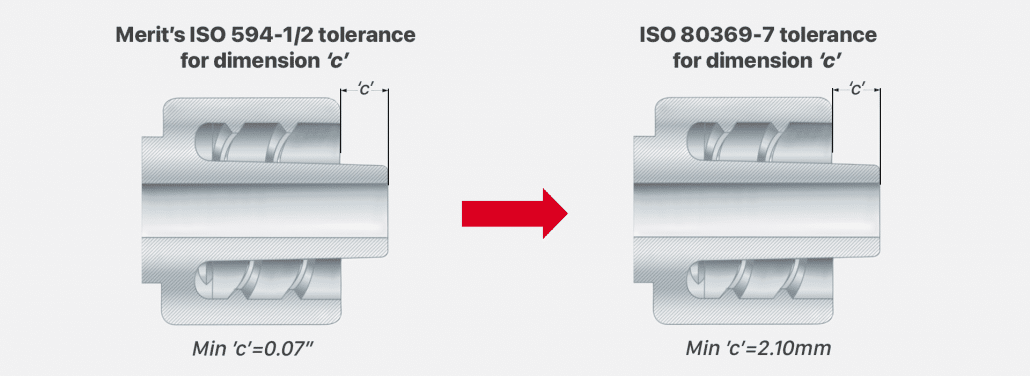
Dimension ‘c’ is another area of Merit’s male luer that will be modified for 80369-7 compliance. Required for compliance, dimension ‘c’ measures the distance between the male luer face to the end of the nozzle. Merit’s current tolerance for dimension “c” is Min ‘c’= 0.07”. The tolerance for dimension ‘c’ will be changed according to the standard to be Min ‘c’ = 2.10mm.
Female Luer
The ANSI/HIMA MD70.1 style thread design was used on some of Merit’s products featuring a female lock luer connection. This thread design does not meet the required ’N’ dimension in the standard. The ANSII/HIMA MD70.1 style thread is too wide at the root to comply with the max ’N’ dimension. Merit will change female lock luers with the ANSII/HIMA MD70.1 style thread with either the 80369-7 double thread or 80369-7 single thread design.

Work with a partner, not just another supplier.
Merit’s cross-functional compliance team is working diligently on this extensive project. Progress on transitioning Merit’s products over to the new luer standard is well underway. Part numbers included in this project come from multiple product families, such as Molded Parts & Components, Valves, Syringes, Tubing Extensions, Percutaneous Access, Fluid Management, Waste Management & Safety Products, and Catheters.
For more information about Merit’s products and our 80369-7 compliance project, contact your Merit business development manager or Merit’s dedicated OEM customer representative at oemorder@merit.com. Request a virtual meeting with Merit’s Product Development Team and get your project started today.
